Scientists (Grigorios Megariotis, Georgios Mikaelian, Haralambos Sarimveis) from the School of Chemical Engineering from National Technical University of Athens (NTUA), were lead- and co-authoring in the following publication:
Grigorios Megariotis, Georgios Mikaelian, Haralambos Sarimveis, In silico investigation of complexes formed between chemical UV filters and cyclodextrins in water, Journal of Molecular Graphics and Modelling, Volume 144, 2026, 109322, https://doi.org/10.1016/j.jmgm.2026.109322.
In this study, Megariotis, Mikaelian and Sarimveis present a detailed computational investigation of supramolecular complexes formed between two widely used chemical UV filters—avobenzone and octocrylene—and the cyclodextrins β-cyclodextrin (β-CD) and 2-hydroxypropyl-β-cyclodextrin (HP-β-CD) in water. The work addresses a central challenge in sunscreen formulation: while chemical UV filters provide efficient UV-A and UV-B protection, they may suffer from photodegradation, limited aqueous solubility, and potential permeation beyond the desired skin layers. Cyclodextrins, owing to their hydrophobic cavity and hydrophilic exterior, are promising host molecules capable of forming stabilizing inclusion complexes.
As stated by the authors, “the main goal of this study is to examine in detail the complexes from a nanoscopic point of view, as well as the complexation process itself, paying particular attention to the thermodynamic description and the stability of the formed supramolecular complexes”. To achieve this, the systems were investigated using long-timescale all-atom molecular dynamics simulations in explicit water. Importantly, each simulation was initiated from an unbound configuration in order “to determine whether noncovalent complexation is a spontaneous process”. In all cases examined, spontaneous inclusion occurred within the simulation timescale, and the resulting complexes remained stable thereafter.
Structurally, both UV filters were found to insert into the cyclodextrin cavity rather than associating externally. The hydrophobic parts of the guest molecules preferentially occupy the interior of the host, while polar groups remain solvent-exposed, thereby optimizing solvation. The simulations further demonstrate that complex formation is accompanied by the displacement of water molecules originally residing inside the cyclodextrin cavity. This release of confined water contributes significantly to the driving force of complexation and reflects a classic hydrophobic inclusion mechanism. Thermodynamically, the calculated binding Gibbs energies are negative for all systems, confirming favourable complex formation. The authors show that van der Waals interactions provide the dominant stabilizing contribution, while electrostatic terms are less favourable. An important aspect of the analysis is the explicit consideration of cyclodextrin strain energy upon binding, highlighting that structural adaptation of the host can partially offset otherwise favorable interactions. Among the systems investigated, the octocrylene–β-CD complex emerges as particularly stable, consistent with the higher hydrophobicity of octocrylene.
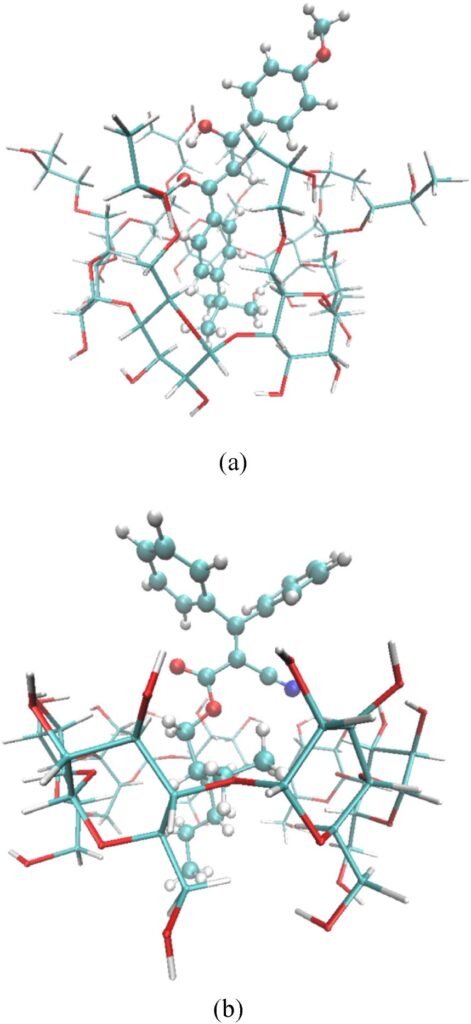
Visualization of complexes formed between UV filters and cyclodextrins: (a) avobenzone (enol form) – HP-β-CD and (b) octocrylene – β-CD. (taken from publication)
A distinctive feature of the study is the analysis of avobenzone tautomerism. Because avobenzone contains a β-diketone moiety, both keto and enol forms were considered. The authors note that the enol form plays a key role in photoprotection, and their simulations confirm that the intramolecular hydrogen bond characteristic of the chelated enol structure remains stable upon complexation. From a thermodynamic perspective, complexation does not destabilize this configuration, an important finding for maintaining UV-A protection efficiency. Beyond binding energetics, the work also evaluates dynamic properties. The calculated self-diffusion coefficients reveal that, upon complexation, the mobility of the UV filters decreases substantially and approaches that of the larger cyclodextrin hosts.
This reduced diffusivity suggests a more constrained behavior in aqueous environments, which may have beneficial implications for formulation stability and prolonged functional performance. Surface analyses further support the inclusion mechanism. The solvent-accessible surface area decreases markedly upon complex formation, particularly for nonpolar regions of the UV filters. This indicates effective shielding of hydrophobic groups within the cyclodextrin cavity and a concomitant enhancement of apparent solubility in water. In this context, the authors conclude that “cyclodextrins do act as an effective protective shield for the two UV filters against surrounding water molecules”.
This work directly reflects the objectives of the PINK project, which promotes the integration of predictive digital tools into Safe-and-Sustainable-by-Design (SSbD) innovation processes. By applying advanced molecular simulations to quantify stability, molecular interactions, solvation behavior and dynamic properties at early research stages, the study demonstrates how computational approaches can generate mechanistic insight before extensive experimental testing. Such digitally supported, mechanistically grounded evaluation of chemical performance aligns closely with PINK’s ambition to enable informed, sustainability-oriented decision-making in the design of safer and more effective chemical products.
Follow this link to read the full publication.
Parts of the research of this work (NTUA) has been funded by the European Union`s R&I project PINK (grant agreement # 101137809).